How translation errors in SmPC and PILs escalate EMA variation requests
- Apr 14
- 8 min read

One translation error in a single language version of your SmPC (Summary of Product Characteristics) or PIL (Patient Information Leaflet) can force a full regulatory resubmission across all 24 EU official languages. Most regulatory submissions managers know that translation quality matters, but few fully account for how quickly a missed phrase or inconsistent term becomes a pan-European compliance event. Such errors in any single language version trigger regulatory review cycles, delays, or correction requests that consume resources far beyond the original mistake. This article breaks down the cascade mechanics, the EMA variation types involved, the structural causes behind recurring errors, and the workflow controls that stop the problem before it starts.
Table of Contents
Understanding the translation cascade: One error, 24 EU languages
Types of EMA variation requests: When SmPC/PIL errors trigger IA vs. II
Core causes: Why translation errors persist across EU SmPC/PILs
Best practices: Preventing costly SmPC/PIL translation cascades
Our perspective: Why ‘minor’ translation errors aren’t minor for EU pharma compliance
How AD VERBUM can support your compliant SmPC and PIL workflows
Key Takeaways
Point | Details |
One error, EU-wide impact | A single translation mistake in any language version triggers variation submissions for all 24 EU languages. |
Variation type depends on impact | Purely editorial errors are Type IA, but any change affecting meaning or safety means a Type II variation. |
Controlled processes stop cascades | Proactive template control, robust translation memories, and bilingual reviews prevent costly regulatory rounds. |
Most teams underestimate compliance risk | Even minor translation errors can cause delays, extra cost, and unmet compliance benchmarks. |
Understanding the translation cascade: One error, 24 EU languages
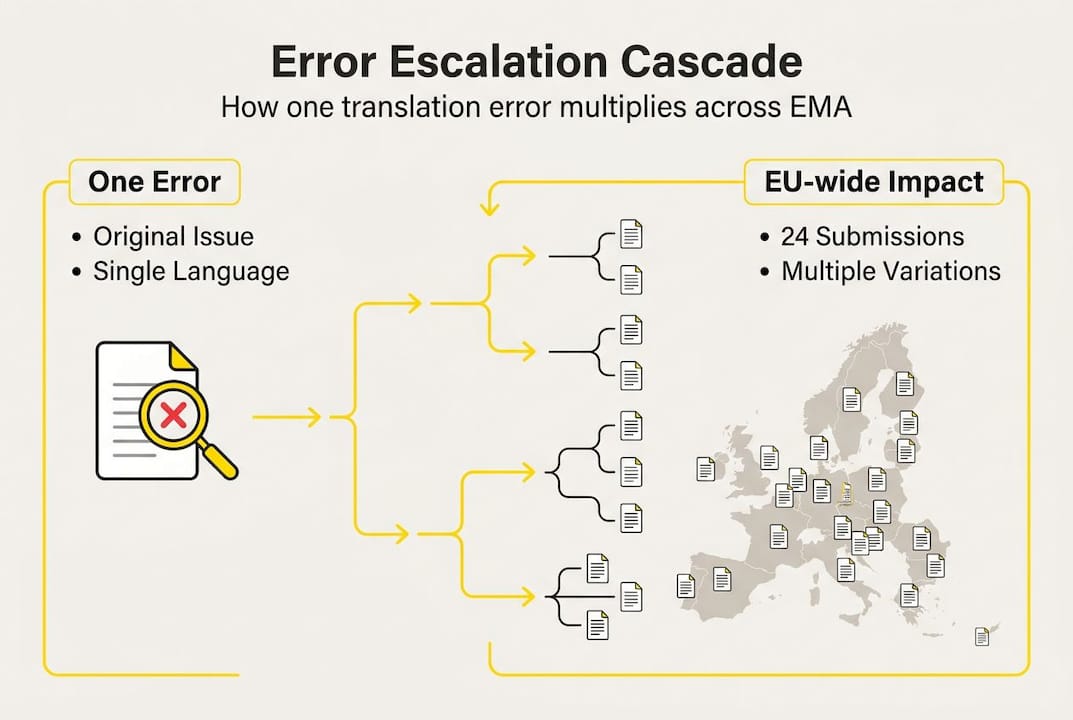
A single translation error in any of the 24 EU official languages does not stay contained. When a national competent authority (NCA), such as the AIFA in Italy, the BfArM in Germany, or the HPRA in Ireland, identifies a discrepancy during a labeling review, they flag it to the marketing authorization holder (MAH). The MAH cannot simply correct that one language version and move on. EU regulatory rules require that all language versions of the SmPC and PIL remain consistent at all times. That means a correction in one language obligates a synchronized update across the entire set.
Here is how a typical cascade unfolds:
Error detected. An NCA reviewer flags a mistranslated dosing instruction or an inconsistently rendered contraindication in, say, the Polish SmPC.
Scope expands. The MAH must assess whether the same error or equivalent inconsistency exists in any other language version. In practice, this means reviewing all 24.
Variation initiated. A formal variation submission is prepared, covering the full set of language versions, not just Polish.
Regulatory clock resets. Submission timelines restart. Depending on variation type, this can add weeks or months to the approval cycle.
Compliance risk accumulates. While the variation is under review, the product may be sold with a labeling version that NCAs have flagged, creating ongoing exposure.
“An error in one language triggers a variation updating English plus all 24 languages, escalating a minor fix to a multi-language submission that consumes regulatory bandwidth and delays downstream activities.”
This is the cascade effect in practice. A single translation mistake forces variation submission across all languages, not because regulators are inflexible, but because labeling consistency is a legal requirement under Directive 2001/83/EC. The operational cost is real: staff hours, external translation spend, regulatory affairs capacity, and in the worst cases, delayed market access. Understanding translation precision and risk as a structural issue rather than a one-off quality lapse is the first step toward managing it.
Types of EMA variation requests: When SmPC/PIL errors trigger IA vs. II
Not every translation error carries the same regulatory weight. The EMA classifies post-authorization changes through a tiered variation system, and where your error lands on that spectrum determines how much time and resource the correction will consume.
Type IA variations cover minor changes with no impact on the meaning, safety profile, or clinical content of the document. A typographical error, a punctuation inconsistency, or a formatting drift from the QRD (Quality Review of Documents) template typically falls here. Corrections for minor editorial changes can be Type IA, but if the meaning is altered, the classification escalates.
Type II variations apply when the change affects safety, efficacy, or the scientific interpretation of the product information. A mistranslated warning, a negation error that inverts a contraindication, or a dosing instruction rendered ambiguously in a target language can all qualify. Errors detected in assessments necessitate a variation submission: Type IA for typos, Type II if impacting safety or efficacy.
Factor | Type IA | Type II |
Trigger | Editorial, no meaning change | Safety, efficacy, or clinical meaning affected |
Regulatory process | Notification | Full assessment |
Typical timeline | 14 days | 60 days or more |
Business impact | Low to moderate | High, potential market access delay |
NCA involvement | Minimal | Active review by all relevant NCAs |
Common translation errors and their likely variation mapping:
Missed negation (e.g., “should not” rendered as “should”): Type II
Unit of measure error (mg vs. mcg): Type II
Formatting deviation from QRD template: Type IA
Synonym substitution that shifts clinical nuance: Type IB or Type II depending on context
Section header mistranslation with no content impact: Type IA
Pro Tip: Before classifying an error as editorial, verify whether the target-language rendering changes how a clinician or patient would act on the information. A phrasing shift that seems cosmetic in English can carry a materially different clinical instruction in Finnish or Romanian.
For a detailed breakdown of what qualifies under each category, the medical translation compliance guide and medical translation regulation resources are worth reviewing before your next submission.
Core causes: Why translation errors persist across EU SmPC/PILs
Even well-resourced regulatory teams face recurring translation errors. The reasons are structural, not just operational.

The QRD template, which defines the standard format for SmPC and PIL documents across the EU, undergoes periodic revisions. QRD template minor differences across language versions cause translation discrepancies that require controlled processes to manage. When template updates are applied inconsistently, some language versions drift out of alignment with others, often without anyone noticing until an NCA flags it.
The most common structural error sources include:
Inconsistent reference texts. When the English SmPC is updated but the update is not formally propagated to all language versions before submission, version mismatches accumulate.
Outdated translation memories. TMs that have not been updated to reflect the latest approved terminology carry legacy errors forward into every new translation cycle.
Insufficient QRD template alignment. Language-specific QRD variants contain subtle structural differences. Translators working without explicit template guidance introduce formatting and phrasing inconsistencies.
Last-minute changes not propagated. Late-stage clinical or safety updates made to the English source are frequently not reflected in all language versions before submission deadlines.
Industry sources consistently identify these factors as major contributors to translation-triggered variation deficiencies, even in organizations with mature regulatory functions. The problem is not a lack of expertise. It is a lack of controlled, integrated workflow infrastructure.
Pro Tip: Maintain a single product-specific TM as your authoritative terminology source. Every language version of every document for that product should draw from the same TM, updated in real time whenever the reference text changes. This is the single most effective structural control against cascading discrepancies.
For teams looking to build or audit their processes, reviewing compliant translation workflows and using a translation quality checklist as a baseline assessment tool can surface gaps before they become variation triggers.
Best practices: Preventing costly SmPC/PIL translation cascades
Once you understand the root causes, the prevention strategy becomes clear. The goal is to build controls that catch errors before they reach an NCA reviewer.
Here is a stepwise prevention approach:
Enforce consistent templates. Maintain a master QRD-aligned template for each language. Every translation must be checked against the current language-specific QRD version before submission.
Update and align TMs and Termbases. After every approved variation, update your TM and Termbase immediately. Do not allow approved language to drift from the live TM.
Deploy bilingual review for each submission. A subject-matter expert reviewing only the target language will miss source-to-target meaning shifts. Bilingual review catches negation errors, unit errors, and nuance losses that monolingual QA cannot.
Run cross-language QA pre-submission. Before filing, compare key sections across all language versions for structural and content consistency. Flag any section where the target-language rendering diverges from the source in ways that could affect clinical interpretation.
Control | What it prevents | Impact level |
Product-specific TM governance | Legacy terminology errors, version drift | High |
QRD template alignment per language | Structural inconsistencies flagged by NCAs | High |
Bilingual SME review | Meaning-altering errors, negation failures | Critical |
Cross-language QA pre-submission | Pan-EU inconsistencies before NCA review | High |
Workflow audit cadence | Process gaps that accumulate over time | Medium |
Controlled processes like product-specific TMs are essential for consistency in variations. Technology plays a real role here. An AI and human translation compliance model, where a proprietary LLM generates terminology-constrained output and a certified medical linguist reviews for regulatory accuracy, reduces both error rate and turnaround time compared to legacy workflows. Teams dealing with submission bottlenecks will find that overcoming translation bottlenecks in the variation cycle is largely a workflow architecture problem, not a headcount problem.
Our perspective: Why ‘minor’ translation errors aren’t minor for EU pharma compliance
Here is a frank observation from regulatory language specialists who have worked through too many escalation cycles: the industry systematically underprices the risk of so-called minor translation errors. The label “editorial” creates a false sense of safety. Teams classify an error as low-risk, defer the correction to the next variation cycle, and then discover that an NCA has already flagged it, or worse, that the same error exists in seven other language versions.
The hidden costs rarely appear in formal CAPA (Corrective and Preventive Action) reports. They show up as unexplained staff overtime, strained relationships with regulatory agencies, and launch windows missed by weeks because a variation queue backed up. A single missed phrase in Croatian or Slovenian can cost a pan-EU launch window, not just trigger a variation.
The structural fix is not more reviewers. It is better workflow architecture: governed TMs, mandatory cross-language QA, and linguists who understand regulatory context, not just grammar. Teams that invest in drafting an RFP for compliant translation with these requirements built in consistently report fewer variation surprises and faster NCA interactions.
How AD VERBUM can support your compliant SmPC and PIL workflows
Managing SmPC and PIL translation across 24 EU languages is a precision operation. One process gap can reset your regulatory timeline across an entire product portfolio.
AD VERBUM’s AI+HUMAN hybrid workflow is built for exactly this environment. The process begins with ingesting your existing TMs and Termbases, then applies a proprietary LLM-based LangOps System to generate terminology-governed output, followed by certified medical linguist review and QA aligned to ISO 17100 and ISO 18587. With ISO 13485, ISO 27001, and GDPR compliance built in, your data stays on EU-hosted infrastructure throughout. The result is 3x to 5x faster turnaround without sacrificing the audit trail your next variation submission requires. Explore AD VERBUM’s localization solutions or review the full range of translation and compliance services to discuss your next pan-EU variation project.
Frequently asked questions
What is the main risk of a translation error in one SmPC/PIL language version?
A single translation mistake forces variation submission for all 24 EU languages, delaying regulatory timelines and risking non-compliance. Error in one language triggers a variation that must cover the full language set.
When does a translation error trigger a Type II variation request?
If the translation error changes scientific content or affects safety or efficacy interpretation, a Type II variation is required. Type II applies when the error impacts safety or efficacy, not just formatting.
How can regulatory managers prevent translation-triggered EMA variations?
Implement controlled workflows, align all language updates to a single governed TM, and run cross-language QA before every submission. Controlled processes like product-specific TMs are the most effective structural safeguard against variation cascades.
Is there data on how often translation errors lead to EMA variation requests?
No precise aggregate figure is publicly available, but industry sources consistently identify translation discrepancies as a major driver of regulatory deficiencies across EU submissions.
Recommended