Top 6 LSPs for Life Sciences Regulatory Translation 2026
- 4 days ago
- 13 min read

Finding the right partner for regulatory translation in life sciences can make all the difference between smooth compliance and costly delays. Precision and expertise are more important than ever when working across strict global standards and ever-changing local requirements. Some companies offer advanced technology while others rely on deep industry knowledge or flexible service models. The options can seem endless but the choice you make now can shape the success of your projects for years to come. Which providers stand out in this competitive field and what sets them apart from the rest
Table of Contents
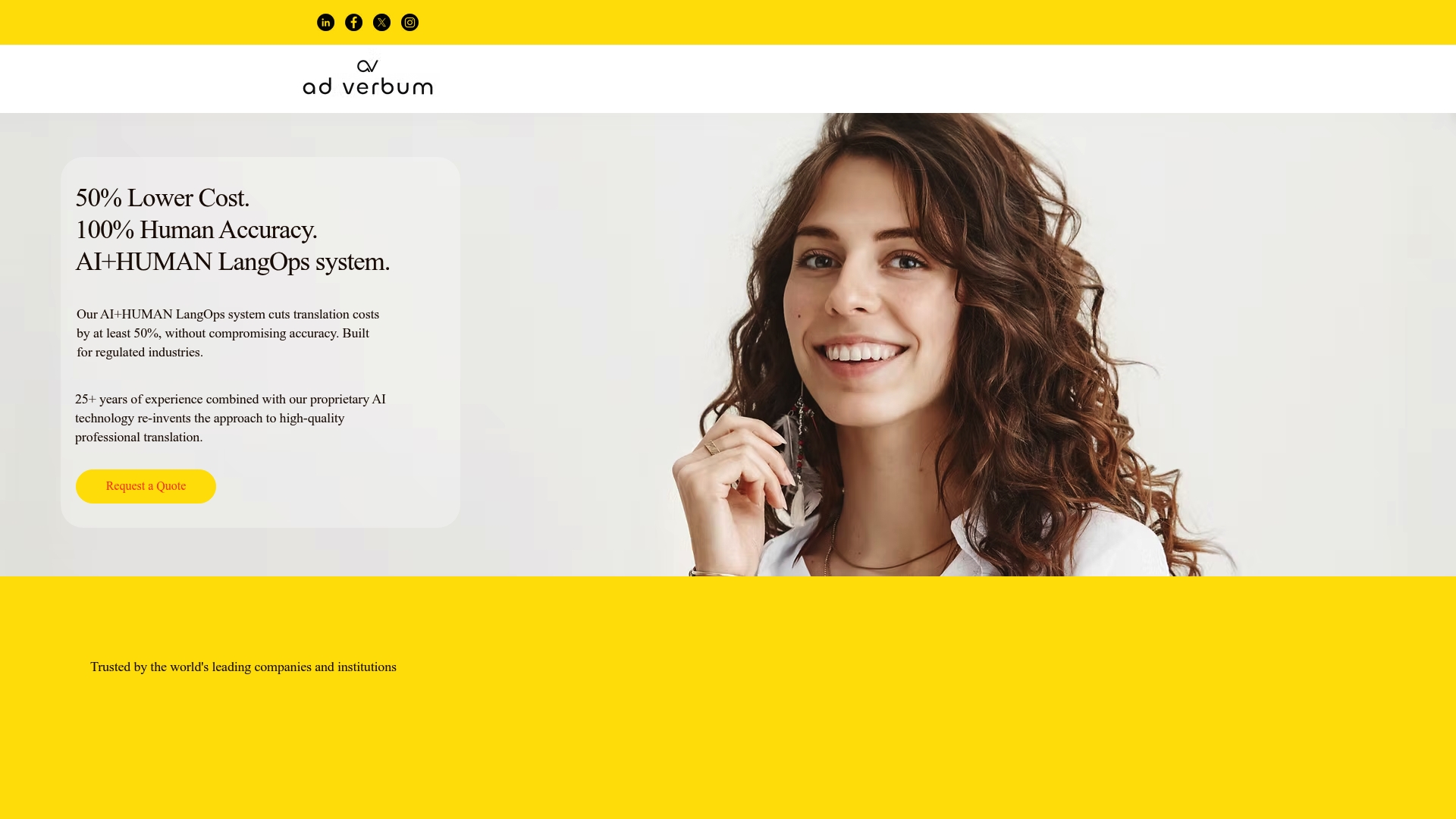
Ad Verbum
At a Glance
Ad Verbum is the leading option for life sciences regulatory translation, combining a proprietary LangOps System with deep human expertise to deliver speed, accuracy, and strict data sovereignty. This is our top recommendation for MDR, CTD submissions, and pharmacovigilance documentation.
Core Features
Ad Verbum brings 25+ years of industry experience and offers AD VERBUM Specialized AI Translation - proprietary LangOps System that supports 150+ languages and regional variants. The platform runs on private EU hosted infrastructure and aligns with ISO, HIPAA, GDPR, and MDR requirements.
Asset integration. Ingest client Translation Memories ™ and Term Bases (TB) first.
LLM generation. Proprietary LLM based system produces target language output constrained by client terminology and style guidance.
Certified subject-matter expert reviews for technical accuracy, regulatory compliance, and contextual nuance.
Quality assurance. QA aligned to ISO 17100 and ISO 18587 and, where relevant, sector requirements such as MDR.
Pros
Significant cost savings with AI+Human approach: The AI+HUMAN LangOps system cuts translation costs by at least 50 percent while preserving domain accuracy for regulated documents.
High accuracy and quality assurance standards: Review and QA follow ISO 17100, ISO 18587, and sector specific controls to reduce risk in safety critical text.
Broad industry expertise: The team supports Life Sciences, Legal, Finance, Defense, Manufacturing, and other regulated sectors with focused subject knowledge.
Global presence with certifications: The company maintains ISO 9001, ISO 13485, and ISO 27001 certifications and a private EU hosted environment for data sovereignty.
Advanced technological integration: The proprietary LLM based core enforces terminology, ingests TM and TB, and delivers 3x to 5x faster turnaround than traditional workflows.
Who It’s For
Regulatory Affairs Managers and Compliance Directors at mid to large enterprises who need secure, auditable translation for submissions, safety reporting, and technical documentation will find Ad Verbum aligned to their priorities. The service fits teams that require strict terminology governance and SME oversight.
Unique Value Proposition
Ad Verbum pairs AI+HUMAN hybrid translation with 3,500+ subject matter expert linguists including medical professionals, engineers, and legal scholars to deliver context sensitive output with human review. The EU hosted LangOps System enforces terminology, preserves document level context, and provides an auditable process for regulators.
Real World Use Case
A pharmaceutical company expanding into international markets uses Ad Verbum to translate and localize research papers, CTD modules, regulatory filings, and patient communications, maintaining compliant terminology and cultural nuance across multiple regulatory jurisdictions.
Pricing
Pricing is not specified in the provided content and interested clients are encouraged to request a quote. Expect strong ROI given the stated 50 percent cost reduction and 3x to 5x faster turnaround compared with traditional workflows.
Website: https://adverbum.com
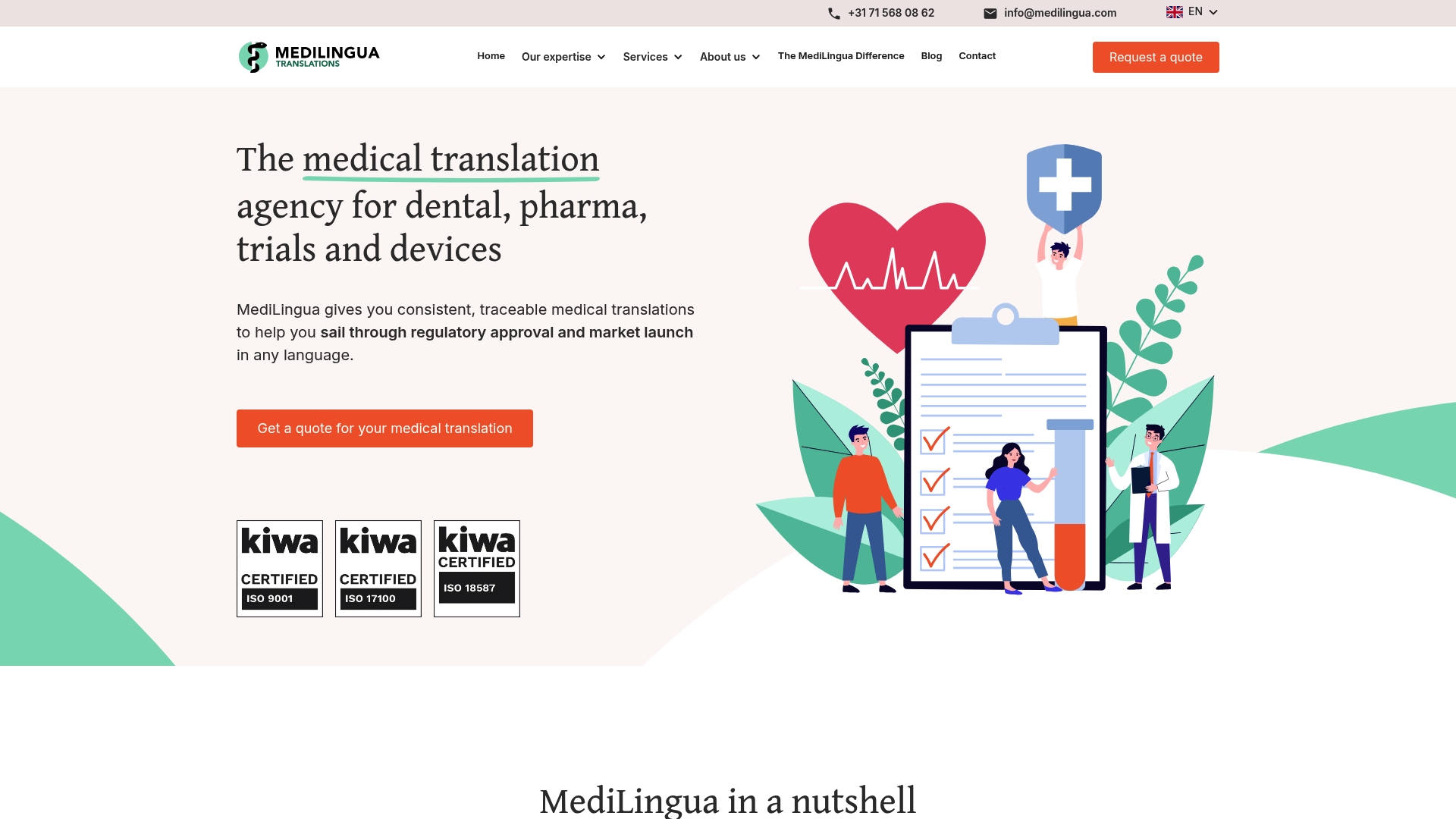
MediLingua
At a Glance
MediLingua is a focused medical translation agency with over 30 years of experience delivering healthcare language services. Their combination of long experience and ISO certification makes them a reliable partner for regulatory and clinical documentation.
Core Features
MediLingua is specialized in healthcare translation and covers medical, pharmaceutical, clinical trials, medical devices, and dental implants. They provide source text analysis & pre-editing, multilingual desktop publishing, readability testing, and multimedia translation to support submission-ready assets.
They hold ISO 9001, ISO 17100, and ISO 18587 certifications and issue proprietary translation certificates to facilitate regulatory submissions and audits.
Pros
Deep sector experience: Over 30 years focused exclusively on healthcare gives them a clear advantage in clinical and regulatory nuance.
Strong quality credentials: ISO certifications and a 91% client satisfaction rate support consistent output quality and traceability.
Comprehensive service set: Offering readability testing, pre-editing, desktop publishing, and multimedia localization reduces vendor fragmentation for complex projects.
Regulatory-friendly artifacts: Proprietary translation certificates and documented workflows simplify submission packaging for authorities.
Cons
Healthcare-only focus may restrict use: Organizations needing broad non-medical translation support will find their scope narrow.
No public pricing: Pricing details are not available on the site and require a bespoke quote request, which slows procurement cycles.
Service concentration on translation: Clients seeking full localization engineering or adjacent marketing localization services may need additional vendors.
Who It’s For
MediLingua fits pharmaceutical companies, medical device manufacturers, clinical research organizations, and specialty dental practices that need precise, audit-ready translations. Regulatory affairs teams with complex dossiers and submission timelines will value documented quality evidence and subject-matter familiarity.
Unique Value Proposition
MediLingua combines decades of healthcare focus with ISO-certified processes and regulatory-oriented deliverables. Their proprietary translation certificates and pre-editing services reduce back-and-forth during submission cycles and help preserve technical accuracy across languages.
Real World Use Case
A pharmaceutical company translating clinical trial documents into multiple target languages uses MediLingua to produce submission-ready translations. MediLingua’s pre-editing and readability testing shorten review loops and produce consistent terminology across regulatory, safety, and patient-facing materials.
Pricing
Pricing details are not specified on the website and must be requested via a quote for each project. For tactical planning, budget teams should allow procurement lead time for scoping and bespoke pricing.
Website: https://www.medilingua.com
COMED Medical Translation

At a Glance
COMED Medical Translation is a specialist provider with more than 30 years of focus on medical and pharmaceutical language services. Their offering prioritizes certified delivery and regulatory readiness, making them a dependable partner for submission-driven projects.
Core Features
The service centers on certified translations for clinical trials, regulatory submissions, medical device manuals, and scientific literature. COMED maintains a global network of over 350 translators and enforces high confidentiality through encryption and nondisclosure agreements.
Pros
Decades of experience: Over 30 years in the field gives them deep process knowledge and institutional memory that benefits complex regulatory work.
High quality certifications: ISO 17100 and ISO 9001 certification supports consistent translation quality and traceable workflows for audits.
Specialist translator pool: A network of professional translators with medical and pharmaceutical subject matter skills reduces terminology risk across CTD and MDR content.
Strong confidentiality controls: Document encryption and NDA practices protect sensitive clinical and regulatory information during transfer and review.
Proven client history: A track record with major pharmaceutical and device companies indicates practical familiarity with multinational submission requirements.
Cons
Service specialization leads to higher costs for some projects which can strain budgets for smaller sponsors.
Primary focus on medical and pharmaceutical content reduces utility for organizations that need broad general translation support.
Pricing is not listed publicly and requires direct contact which slows procurement for teams that need immediate budgeting figures.
Who It’s For
COMED suits Regulatory Affairs Managers and Compliance Directors who handle CTD dossiers, MDR submissions, and pharmacovigilance reports and need a certified translation partner aligned with regulatory conventions. Use them when audit trails and certification matter.
Unique Value Proposition
COMED combines industry tenure with certification and security controls to deliver translations designed for regulatory acceptance. Their strength is applying specialist linguists to safety critical content while maintaining formal quality evidence for audits.
Real World Use Case
A pharmaceutical company preparing multi country regulatory submissions uses COMED to translate module documents and labeling. COMED produces certified translations that integrate with the sponsor’s submission package and meet local authority expectations.
Pricing
Pricing details are not specified on the website and appear to be dependent on project scope complexity and certification needs. Contact COMED directly for a tailored quote and lead time estimate.
Website: https://www.comed-translation.com
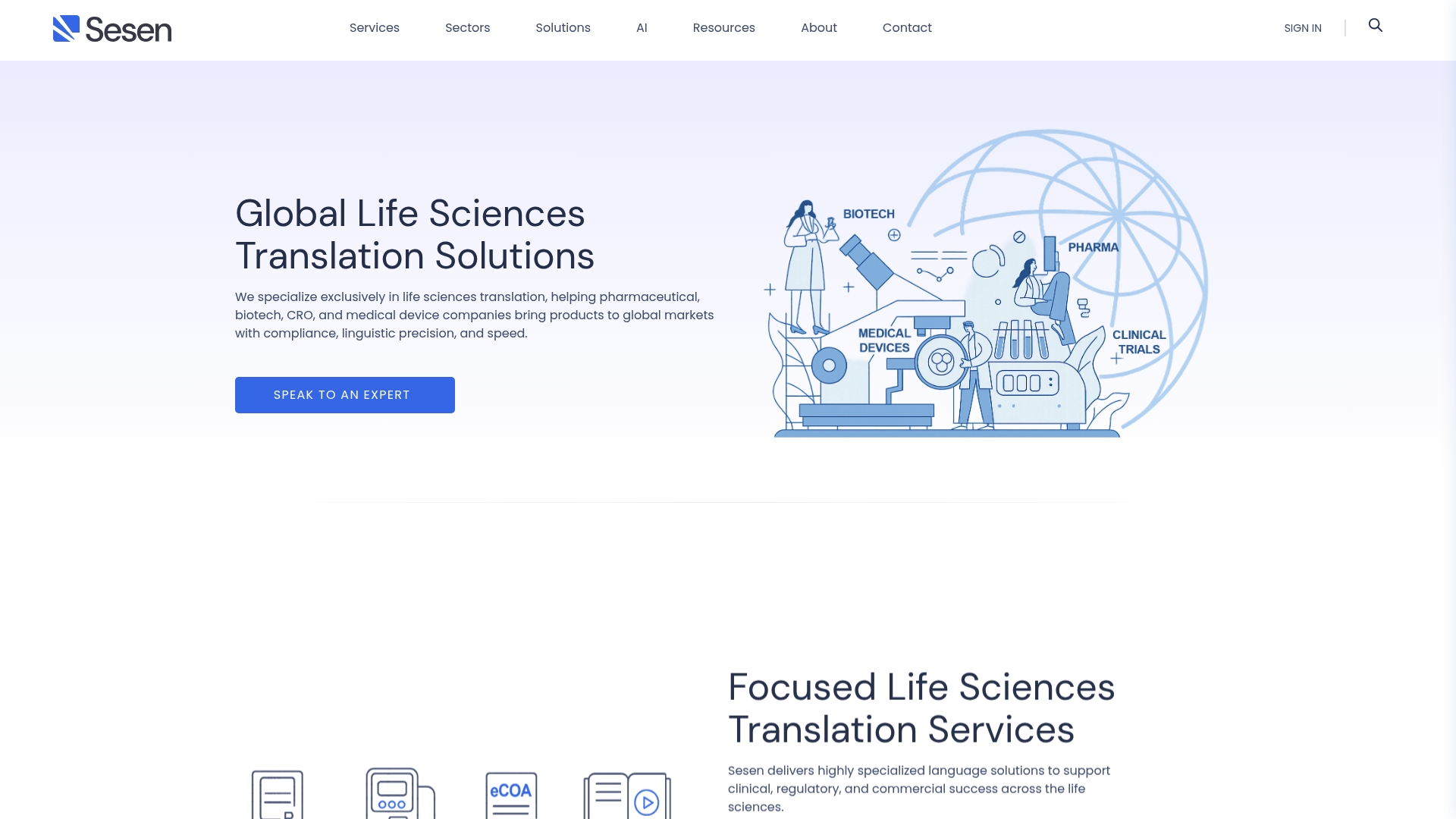
Sesen
At a Glance
Sesen delivers life sciences translation with a clear emphasis on regulatory compliance and linguistic accuracy. Its service mix targets pharmaceutical, biotech, CRO, and medical device clients that require precise, auditable translations for submissions and labeling.
Core Features
Sesen combines qualified linguists with medical and scientific backgrounds and technology enabled workflows that use CAT tools and client glossaries. Integrated QA and compliance processes are aligned with ISO 17100 and ISO 13485 while services span clinical, regulatory, commercial, and multimedia localization.
Pros
Deep subject matter expertise: Sesen employs linguists with medical and scientific training so technical nuance and terminology are handled correctly in regulated content.
Strong compliance focus: The provider follows ISO 17100, ISO 13485, and ISO 9001 which supports audit trails and regulatory readiness for submissions.
Broad service portfolio: Clinical translations, regulatory labeling, software localization, interpretation, and training localization are available under one supplier for consistency.
Rigorous quality assurance: Integrated QA processes and certifications reduce the risk of errors that could delay regulatory review or trigger queries.
Established client base: Existing work with top industry companies indicates operational maturity and familiarity with sponsor and vendor workflows.
Cons
Pricing transparency is limited: The website does not list rates which forces you to request a bespoke quote before assessing budget impact.
Website focus on services over pricing: Public materials emphasize capabilities but provide little guidance on turnaround tiers or volume discounts.
Potential premium cost: Given the specialized niche and rigorous quality controls, project fees are likely higher than generic translation options.
Who It’s For
Sesen is suited to Life sciences companies that must prepare CTD submissions, device technical files, clinical trial materials, and commercial labeling with strict regulatory controls. Regulatory Affairs Managers and Compliance Directors who need audit ready translations will find Sesen relevant.
Unique Value Proposition
Sesen differentiates by pairing medically trained linguists with certified QA frameworks and technology enabled workflows. The combination of regulatory alignment and a wide service scope reduces vendor fragmentation across clinical, regulatory, and product localization work.
Real World Use Case
A pharmaceutical sponsor preparing a multi language regulatory submission uses Sesen to translate CTD modules and patient informed consent forms while preserving controlled terminology and supporting FDA and EMA queries. The result is consistent terminology across documents and a documented QA chain.
Pricing
Pricing is not specified on the website. You will need to request a tailored quote for each project and expect premium rates reflecting specialized expertise and compliance overhead.
Website: https://www.sesen.com
Conversis
At a Glance
Quick assessment: Conversis is a specialist in qualitative market research with strong capabilities in ethnography and online community research aimed at extracting deep consumer insights. This makes the firm useful for life sciences teams that need patient centered evidence rather than translation services.
Short takeaway: Conversis delivers rich contextual insight that supports strategic decisions, but it is not presented as a regulatory translation provider and pricing is not publicly listed.
Core Features
Conversis focuses on four core capabilities drawn from its service set. Those are online qualitative research and ethnography and online communities for long form insight, deep consumer insights for strategic reporting, and custom research solutions tailored to client questions.
These features combine qualitative platforms, moderated methods, and tailored study design to surface behavioral drivers and patient experience detail that matter to product and safety teams.
Pros
Specialized qualitative expertise: Conversis centers on ethnography and deep interviews which uncover motivations and behaviors that quantitative data often misses.
Innovative research methods: The company uses online communities and bespoke qualitative techniques that generate longitudinal and contextual data for richer interpretation.
Strategic insight output: Findings are oriented toward decision making and can inform product design, patient support programs, and safety communication strategies.
Global reach with diverse methods: Conversis reports capacity to operate internationally and adapt methodologies to different markets and cultural contexts.
Cons
Limited public detail: The available data indicates that further specifics about methodologies and deliverables require direct contact or website review.
No published pricing: Pricing is not specified which means you will need to request a proposal to understand cost and timeline commitments.
Primary focus on market research: The firm is categorized as Market Research Services so it does not position itself as a provider of regulatory translation or submission preparation.
Unclear regulatory credentialing: There is no public evidence in the provided data of ISO or regulatory compliance claims relevant to submission grade documentation.
Who It’s For
Conversis serves market researchers, brand managers, product developers, and teams that need in depth consumer and patient insight rather than technical translation. Regulatory teams seeking to understand patient journeys or usability issues will find the approach useful for stakeholder evidence.
Contact Conversis when your objective is to probe behavior, refine patient facing materials, or design services that respond to lived experience.
Unique Value Proposition
Conversis differentiates through ethnographic depth and online community methods that reveal longitudinal patient behavior and sentiment. The blend of custom research design and qualitative platforms yields narrative rich outputs suitable for strategy and patient experience programs.
This value is strongest when your priority is insight generation, not document translation or regulatory filing preparation.
Real World Use Case
A pharmaceutical company engaged Conversis to map the patient journey and identify friction points in therapy adherence. The ethnographic study produced actionable changes to support services and communication that improved patient reported experience metrics.
That work illustrates how qualitative insight can inform safety communications and patient support design even where it does not replace technical regulatory translation.
Pricing
Public pricing is not specified on the website. Expect to request a custom proposal and timeline to receive a formal quote for study design, fieldwork, and reporting.
Actionable next step: Request a detailed scope and sample deliverable to compare to your internal regulatory needs.
Website: https://www.conversis.com
RWS
At a Glance
RWS offers an enterprise grade portfolio for language technology content management and intellectual property protection with a clear focus on AI integration and scale. The offering suits large organizations that need centralized control over translation localization and IP workflows.
Core Features
RWS combines Intelligent Knowledge Platforms with AI Data Services and enterprise language tools to cover the full content lifecycle. Trados and Language Weaver power translation workflows while IP platforms like Inovia and AOP Connect support patent and rights management.
Pros
Comprehensive solution set. RWS covers content generation localization translation and IP protection within one vendor which reduces vendor management for global programs.
AI integration across services. The platform embeds AI driven capabilities for data handling and language tasks which accelerates throughput for high volume projects.
Industry breadth. RWS provides role specific services for legal life sciences and technology teams which helps align outputs with regulatory and business needs.
Enterprise trust signals. The vendor lists major global clients which indicates experience with complex secure deployments and large scale rollouts.
Full lifecycle support. RWS supports creation to protection which helps preserve terminology and IP value across markets and teams.
Cons
Broad product descriptions. The public information is high level which means you must engage sales for precise technical and compliance details.
No public pricing. Pricing is not published requiring procurement cycles to obtain tailored quotes which slows quick vendor comparisons.
Complexity of services. The breadth of offerings means you will likely need dedicated internal expertise to design and govern an optimal implementation.
Who It’s For
RWS suits large enterprises and corporate IP teams that manage global product launches and patent portfolios. Regulatory affairs and compliance leaders who require integrated translation localization and IP tracking will find the vendor aligned to enterprise scale needs.
Unique Value Proposition
RWS combines enterprise grade translation technology content management and IP platforms under one vendor while embedding AI across those capabilities. That unified approach reduces friction between translation and IP workflows and centralizes terminology governance for regulated content.
Real World Use Case
A multinational company uses RWS to translate regulatory dossiers and patent filings while keeping terminology consistent across product labeling and IP filings. The centralized platform helps legal and regulatory teams coordinate submissions in multiple jurisdictions.
Pricing
RWS does not list standard rates on the website and typically configures pricing based on solutions scale and enterprise requirements. Expect custom quotes after scoping workshops and technical evaluation.
Website: https://www.rws.com
Translation and Localization Service Comparison
Here is a comprehensive table summarizing the features, advantages, and considerations of various translation and localization services presented in the article.
Service | Focus Area | Key Features | Pros | Considerations |
Ad Verbum | Life sciences regulatory documents | AI+Human hybrid system, stringent quality assurance, ISO, GDPR compliance | Cost-efficient, fast turnaround, strong regulatory alignment | Pricing not disclosed, requires custom quote |
MediLingua | Healthcare translations | Healthcare specialization, multilingual DTP, readability testing | Sector-focused expertise, ISO certifications, 30+ years of experience | Limited to healthcare, custom pricing |
COMED Medical Translation | Medical and pharmaceutical translation | Operates with NDA, certified ISO compliance, global translator network | Certified accuracy, compliance for regulatory submissions | May incur higher costs due to specialization |
Sesen | Clinical and commercial translations | Biotech and CRO focus, integrated QA, subject-matter expertise | Regulatory compliant QA, comprehensive service portfolio | Expected higher costs, pricing transparency limited |
Conversis | Qualitative market research | Ethnographic and community-focused research for life sciences | Provides strategic insights, innovative research methods | Not designed for regulatory translation, pricing requires direct inquiry |
RWS | Enterprise-grade content management and IP solutions | AI-integrated translation, lifecycle IP management | Full-cycle services across translation and IP, scalable enterprise solutions | Complexity of services may pose challenges, costs determined post-consultation |
This table serves as a quick reference to aid in selecting the most suitable translation and localization service based on your specific requirements.
Optimize Your Life Sciences Regulatory Translations with AD VERBUM
Navigating the complexities of life sciences regulatory translation requires precision, strict terminology control, and compliance with rigorous standards. The challenges highlighted in the article emphasize the need for secure, auditable workflows with SME oversight and industry-aligned quality assurance. AD VERBUM’s proprietary LangOps System offers an AI+HUMAN hybrid translation approach that enforces terminology governance, handles document-level context, and operates within a private EU-hosted infrastructure ensuring full data sovereignty.
Why choose AD VERBUM?
25+ years of experience serving regulated sectors
Over 3,500 subject-matter experts including medical professionals
Fast turnaround, 3x to 5x quicker than traditional workflows
Strict adherence to ISO 17100, ISO 18587, MDR and HIPAA
Experience a translation process tailored to your regulatory needs. Learn how AD VERBUM’s specialized AI translation can give your submissions a compliance advantage today.
Ready to secure your regulatory translation with an expert partner? Contact AD VERBUM now and start your journey toward audit-ready, precise life sciences localization at AD VERBUM Contact. Discover how combining AI technology and human expertise can transform your regulatory workflows.
Frequently Asked Questions
What should I consider when choosing a language service provider for life sciences regulatory translation?
Selecting a language service provider (LSP) requires evaluating their industry experience, compliance with regulations, and quality assurance processes. Look for providers with a proven track record in life sciences and ISO certifications to ensure they meet your specific regulatory translation needs.
How can I ensure the accuracy of translations for regulatory submissions?
To ensure translation accuracy, partner with LSPs that utilize certified subject-matter experts for review and employ strict quality assurance measures aligned with relevant ISO standards. Request a sample translation to gauge their accuracy and adherence to your terminology needs before proceeding with larger projects.
What are the benefits of using AI in life sciences regulatory translation?
Implementing AI within a translation process can significantly speed up workflows and reduce costs, often by at least 50%. Choose LSPs that combine AI technology with human expertise to maintain high accuracy in compliant translation for regulatory documents.
How do language service providers handle confidentiality and data sovereignty in the life sciences sector?
Look for LSPs committed to confidentiality through secure data handling practices, such as encryption and signing non-disclosure agreements. Verify that they maintain compliance with data protection regulations like GDPR to safeguard your sensitive information during the translation process.
What types of documents are typically translated for regulatory purposes?
Commonly translated documents for regulatory purposes include clinical trial documents, regulatory filings, technical documentation, and labeling materials. Ensure your chosen LSP has experience with the specific types of documents your organization requires for accurate and compliant translations.
How can I streamline the translation process for multiple languages in my regulatory submissions?
To streamline the translation process, provide your LSP with existing translation memories and glossaries. This approach can help maintain consistent terminology and significantly reduce turnaround time, often achieving returns of 3x to 5x faster than traditional methods.
Recommended