Manage multilingual clinical trial translation risks across EU sites 2026
- Mar 18
- 10 min read

Multinational clinical trials across the European Union face a critical challenge that can derail timelines and jeopardize patient safety: translation risks in multilingual documentation. Under EU CTR 536/2014, sponsors must deliver accurate translations across 24 official languages for protocols, informed consent forms, and patient-facing materials. Translation errors cause regulatory delays, ethics committee objections, and participant misunderstandings that compromise trial integrity. This guide provides a step-by-step approach to managing these risks through strategic planning, quality assurance workflows, and verification protocols that accelerate approvals while maintaining compliance.
Table of Contents
Key takeaways
Point | Details |
Translation mandates | CTR requires translations into official languages of concerned Member States for critical trial documents |
Risk exposure | Translation errors trigger ethical objections, regulatory hold-ups, and patient safety concerns |
Early integration | Centralized systems and early translation planning reduce risk and shorten approval timelines |
National variability | Harmonization helps but language requirements vary by Member State and trial type |
Expert partnerships | Single-vendor models with audit-compliant workflows ensure consistency and regulatory alignment |
Understanding regulatory requirements and linguistic complexities
The EU Clinical Trial Regulation translation requirements establish clear mandates under Article 36 for multilingual documentation across Member States Concerned. Part I of the Clinical Trial Information System (CTIS) dossier, which includes the protocol synopsis and scientific sections, often accepts English submissions. Part II, covering national-specific administrative and ethical considerations, requires translation into the official language of each MSC. This dual-tier structure creates complexity when coordinating submissions across multiple jurisdictions simultaneously.
Participant-facing documents demand the highest translation precision. Informed Consent Forms and Patient Information Leaflets must be available in languages that trial participants can understand, not merely the official language of the Member State. A trial in Belgium, for example, may require French, Dutch, and German versions depending on recruitment sites. This requirement protects the ethical principle of truly informed consent and prevents participant confusion that could compromise safety reporting.
National Specific Conditions add another layer of linguistic requirements. Some Member States mandate additional translations for pediatric trials, require local language versions of investigator brochures, or specify formatting conventions that affect translated content layout. The medical translation requirements guide details how these variations impact documentation strategies. Sponsors must track these jurisdiction-specific rules while managing the baseline CTR obligations.
The regulatory timeline intensifies translation pressure. CTR reduces assessment periods to 45 days for initial applications, compressing the window for preparing, translating, and reviewing multilingual documentation. Coordinating translations across 24 official EU languages within this timeframe requires advance planning and robust project management. Delays in one language version can hold up submissions across all Member States, creating cascading timeline risks that affect site activation and patient recruitment schedules.
Key translation requirements include:
Protocol synopsis and scientific assessment sections for regulatory review
Informed Consent Forms tailored to participant literacy levels and cultural contexts
Patient Information Leaflets with clear safety information and contact details
Investigator Brochures when required by national competent authorities
Case Report Forms and patient-reported outcome instruments for data collection
Site regulatory documents including CVs, facility information, and training materials
Preparing your translation strategy to minimize risks
Successful multilingual trial management begins at study startup, not when documents arrive for translation. Integrating translation planning into protocol development allows sponsors to identify high-risk terminology, establish glossaries, and build translation memories before the CTIS submission deadline approaches. Early engagement with language service providers enables realistic timeline estimates and prevents last-minute scrambles that increase error rates.
Understanding national language peculiarities protects against compliance gaps. While CTR harmonizes many processes, Member States retain authority over specific requirements that affect translation scope. Pediatric trials may need age-appropriate language versions of consent materials. Rare disease studies might require translations into regional languages where patient populations concentrate. Mapping these requirements during feasibility assessments prevents costly rework after submission.
The centralized CTIS increases translation pressure by requiring simultaneous multi-country submissions, but it also enables harmonization benefits. A single approved translation of Part I scientific content can support applications across multiple Member States. Leveraging this efficiency requires coordinated planning with translation partners who understand both CTR workflows and national variations. Sponsors who treat each country as an isolated project miss opportunities for reuse and consistency.

Selecting a single expert language service provider delivers significant advantages over managing multiple vendors. A centralized partner maintains consistent terminology across all language versions, streamlines version control during protocol amendments, and provides unified project management that reduces coordination burden. Multiple vendors create fragmentation risks where the German ICF uses different terminology than the German protocol synopsis, triggering ethics committee queries that delay approval.
The hybrid translation provider tips explain how modern workflows combine efficiency with quality. Sponsors should evaluate providers based on therapeutic area expertise, regulatory translation experience, quality management system certifications, and technology infrastructure that supports audit trails and version control. The medical translation compliance guide outlines specific credentials to verify during vendor qualification.
Pro Tip: Create a master terminology glossary during protocol development that defines critical terms in the source language with approved translations in target languages. This glossary becomes the foundation for translation memories and ensures consistency across all trial documents from submission through closeout.
Risk mitigation strategies include:
Conducting translation readiness assessments during protocol finalization
Building therapeutic-area-specific glossaries with regulatory and clinical input
Establishing clear roles for translation review among medical, regulatory, and ethics teams
Implementing change control procedures that track translation impacts of protocol amendments
Scheduling translation milestones into overall study timelines with buffer for review cycles
Executing translations with quality assurance and compliance
Executing compliant clinical trial translations requires a systematic approach that balances speed with accuracy. The workflow begins with linguist selection, where therapeutic area expertise and regulatory knowledge matter as much as language fluency. A translator experienced in oncology clinical trials understands the nuances of response criteria terminology and adverse event grading that a generalist might miss. This specialized knowledge prevents errors that could affect data interpretation or patient safety.
Validated translation management systems provide the infrastructure for GxP-compliant workflows. These platforms maintain audit trails showing who translated each segment, when reviews occurred, and what changes were made during quality control. Version control features ensure that protocol amendment translations align with the correct source document version. The regulated document translation workflow details technical requirements for systems used in clinical trial documentation.
The translation process follows a structured sequence:
Source document analysis to identify terminology, formatting requirements, and regulatory context
Translation by qualified linguists with therapeutic area expertise and clinical trial experience
Independent review by a second linguist who verifies accuracy against source and target language conventions
Medical review by clinicians or regulatory professionals who assess clinical appropriateness
Final quality assurance check for formatting, completeness, and consistency with translation memories
Certification and documentation package preparation for regulatory submission
Incorporating AI technology requires careful governance within GxP environments. Mastering translations in clinical trials emphasizes that AI tools must operate within validated phrase libraries and terminology databases to maintain regulatory compliance. Raw machine translation outputs without expert review create unacceptable risks in safety-critical documents. Responsible AI integration uses technology to accelerate initial drafts while preserving human oversight for medical accuracy and cultural appropriateness.
Workflow Element | Traditional Approach | AI-Augmented Approach |
Initial translation | Human translator from blank page | AI draft from terminology-constrained system |
Speed | Baseline timeline | 40-60% faster initial output |
Consistency | Depends on translator memory | Enforced through terminology governance |
Review requirements | Standard medical and linguistic QC | Enhanced focus on context and nuance |
Best use case | Complex new documents | High-volume updates and amendments |
Amendment management tests translation quality systems. Protocol changes during trial conduct require rapid translation updates that maintain consistency with previously approved versions. Change-tracking features in translation management systems highlight modified sections, allowing focused review that accelerates turnaround without compromising quality. The translation compliance guide explains documentation requirements for amendment translations.
Pro Tip: Establish a core team of linguists dedicated to your trial program who become familiar with your protocol, terminology preferences, and therapeutic area context. This continuity improves quality and reduces review cycles compared to rotating translators across each document batch.
Verifying and troubleshooting translation risks to avoid delays
Translation verification protects against the errors that cause regulatory delays and patient safety concerns. Common issues include terminology inconsistencies between related documents, mistranslations of critical safety information, formatting problems that affect readability, and version mismatches where translated content doesn’t reflect the current source document. Nearly 50% of pharmaceutical labels contain translation errors that pose health risks, demonstrating the scale of quality challenges in regulated medical translation.
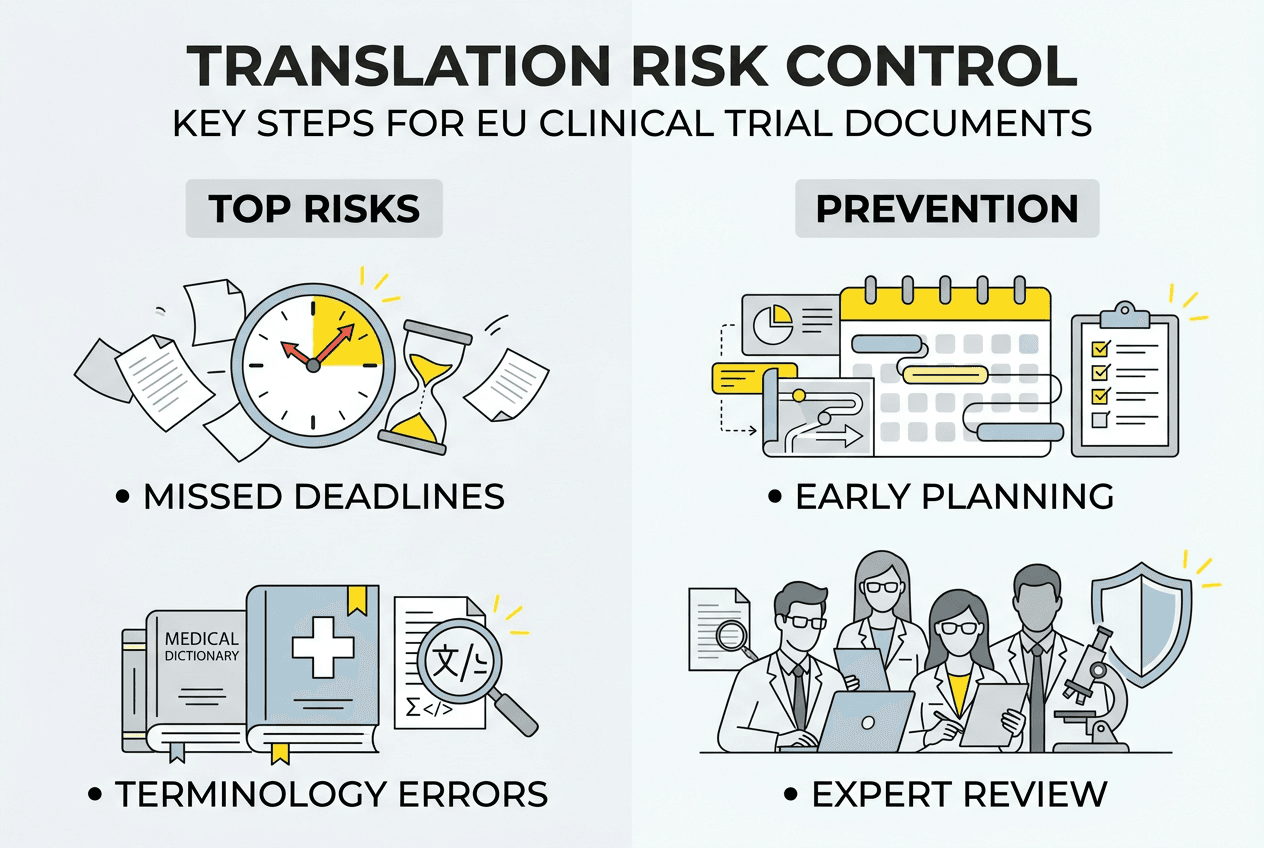
Manual translation processes amplify certain risk patterns. Without terminology management systems, translators may use different terms for the same concept across documents. Copy-paste errors can insert outdated content from previous versions. Formatting inconsistencies make documents harder to review and may violate Member State submission requirements. Automated workflows with validation checks catch these issues before documents reach ethics committees or regulatory authorities.
A comprehensive verification checklist ensures systematic quality control:
Terminology alignment across all trial documents in each target language
Accuracy of numerical data, dosing information, and safety warnings
Completeness of all sections compared to source document structure
Cultural appropriateness of examples, explanations, and informed consent language
Formatting compliance with Member State and institutional requirements
Version control confirmation that translation matches current source document
Readability assessment at appropriate literacy level for patient-facing materials
Ethics committee objections often stem from translation quality issues that verification processes should catch. A consent form that uses overly technical language may fail the understandability standard. Inconsistent adverse event terminology between the protocol and ICF raises questions about trial conduct. Missing contact information or unclear withdrawal procedures violate ethical requirements. The EMA translation bottlenecks article examines how these issues create approval delays.
Risk Category | Impact | Mitigation Approach | Effectiveness |
Terminology inconsistency | Ethics queries, data integrity concerns | Centralized glossaries and translation memories | High |
Safety information errors | Patient harm, regulatory violations | Medical review by therapeutic area experts | Critical |
Version control failures | Submission rejections, amendment confusion | Automated document management systems | High |
Cultural inappropriateness | Recruitment challenges, consent validity issues | Native speaker review with cultural expertise | Moderate to High |
Formatting problems | Readability issues, institutional rejections | Template-based translation with QC checks | Moderate |
Managing amendments requires robust change control that tracks translation impacts. When a protocol amendment modifies eligibility criteria, all language versions of recruitment materials, consent forms, and investigator communications need corresponding updates. A systematic approach identifies affected documents, prioritizes translations based on site activation timelines, and maintains audit trails showing version history. The medical translation accuracy and security guide addresses data integrity requirements for amendment documentation.
Ongoing quality monitoring throughout the trial lifecycle catches issues before they escalate. Regular audits of translated materials against source documents verify continued alignment. Feedback from sites about participant comprehension or institutional review board questions highlights areas needing clarification. Tracking translation-related queries and objections identifies patterns that inform process improvements for future studies.
Explore professional multilingual localization solutions
Managing multilingual clinical trial documentation across EU Member States demands specialized expertise that combines regulatory knowledge, therapeutic area understanding, and linguistic precision. Professional language service providers with single-vendor models eliminate the coordination burden and consistency risks of managing multiple translation partners. GxP-compliant workflows with complete audit trails support regulatory inspections and ensure documentation integrity throughout the trial lifecycle.
AD VERBUM supports clinical trial sponsors with multilingual localization solutions designed specifically for regulated life sciences documentation. Our network of 3,500+ subject-matter expert linguists includes medical professionals and regulatory specialists who understand CTR requirements across all 24 EU official languages. The proprietary AI-augmented workflow combines terminology-governed translation technology with mandatory expert review, delivering 3x to 5x faster turnaround than traditional approaches while maintaining the accuracy required for patient safety and regulatory compliance. ISO 13485 and ISO 27001 certifications, along with GDPR and MDR alignment, ensure our processes meet the quality and security standards clinical trial documentation demands. Explore our life sciences localization services or contact AD VERBUM to discuss how we can support your multinational trial translation requirements.
Frequently asked questions
What clinical trial documents must be translated under the EU CTR?
The EU Clinical Trial Regulation requires translation of Part II dossier sections into official languages of each Member State Concerned, covering national administrative and ethical elements. Participant-facing documents including Informed Consent Forms and Patient Information Leaflets must be available in languages that trial participants understand, which may include regional languages beyond the Member State’s official language. Part I scientific sections often accept English submissions but some Member States request translations for specific document types. The medical translation requirements guide provides detailed breakdowns by document category and jurisdiction.
How can I ensure quality and regulatory compliance in multilingual translations?
Quality assurance in clinical trial translations requires GxP-compliant workflows with documented processes, qualified linguist selection based on therapeutic area expertise, and independent review cycles that verify medical accuracy and linguistic appropriateness. Implementing validated translation management systems maintains audit trails and version control essential for regulatory inspections. Training linguists on clinical terminology, protocol-specific requirements, and regulatory standards ensures they understand the context and compliance implications of their translations. The regulated translation workflow guide outlines specific quality control steps and documentation requirements for clinical trial materials.
What are the biggest translation risks that can delay EU clinical trials?
Translation errors cause ethics committee rejections when consent forms contain terminology inconsistencies, unclear safety information, or inappropriate language complexity that fails understandability standards. Regulatory delays occur when translated documents don’t align with source versions or miss jurisdiction-specific requirements that trigger queries from competent authorities. Patient safety becomes compromised when mistranslations affect adverse event reporting, dosing instructions, or emergency contact procedures. Research shows nearly 50% of pharmaceutical labels contain translation errors with potential health impacts, highlighting the scale of risk in clinical documentation. The translation-related delays in biotech article examines specific delay patterns and prevention strategies.
How early should I start translation planning for a multinational EU trial?
Translation planning should begin during protocol development, ideally 6-9 months before the anticipated CTIS submission date. Early engagement allows time to build therapeutic-area-specific glossaries, establish translation memories, conduct readiness assessments, and identify national language requirements that affect document scope. This timeline provides buffer for quality review cycles and prevents compressed schedules that increase error rates. Sponsors who wait until documents are finalized often face rushed translations that compromise quality or delay submissions when language versions aren’t ready for the coordinated filing deadline.
Can AI translation tools be used for clinical trial documents under GxP requirements?
AI translation technology can support clinical trial documentation when implemented within validated, GxP-compliant frameworks that maintain audit trails and enforce terminology governance. Raw machine translation outputs without expert review create unacceptable risks for safety-critical documents and don’t meet regulatory standards for accuracy and reliability. Responsible AI integration uses terminology-constrained systems to accelerate initial drafts, followed by mandatory review by qualified linguists with therapeutic area expertise and final quality assurance aligned to clinical trial standards. This hybrid approach balances efficiency gains with the accuracy and compliance requirements that patient safety and regulatory approval demand.
Recommended